Address
14 -7311 Minoru Blvd
Richmond, BC, Canada V6Y3P3
Work Hours
Monday to Friday: 7AM - 7PM
Weekend: 10AM - 5PM
Address
14 -7311 Minoru Blvd
Richmond, BC, Canada V6Y3P3
Work Hours
Monday to Friday: 7AM - 7PM
Weekend: 10AM - 5PM

Nature Microbiology: Scientists confirm that an imbalance of intestinal flora promotes liver cancer and that Klebsiella pneumoniae is the causative agent of liver cancer.
About six years ago, American scientists published an unexpected research result in the top journal Cell.
They found that feeding the soluble dietary fiber inulin, pectin or oligofructose to mice whose gut flora was already imbalanced led to the development of primary liver cancer in 40% of the mice [1].
This study suggests that intestinal flora imbalance is closely related to the development of liver cancer. However, it has been unknown whether intestinal microorganisms directly promote the occurrence or development of hepatocellular carcinoma.
Recently, an important paper was published in the prestigious journal Nature Microbiology by the team of Yu Jun from the Chinese University of Hong Kong [1]. Based on fecal samples from liver cancer patients and mouse models, they found that the intestinal microorganisms of liver cancer patients are indeed imbalanced, and upon transplantation of intestinal bacteria from liver cancer patients to mice, this leads to damage of the intestinal barrier and entry of viable bacteria into the liver of mice, which in turn promotes the development of hepatocellular carcinoma.
What’s more, they also demonstrated that Klebsiella pneumoniae in the gut is actually responsible for promoting liver inflammation and liver cancer development in mice, and that it enters the liver to directly interact with liver cancer cells, promoting cell proliferation and activating pro-cancer signaling pathways. In short, Klebsiella pneumoniae in the gut is actually the pathogen that causes liver cancer.

To map the relationship between gut flora and liver cancer, Jun Yu’s team collected feces from patients with hepatocellular carcinoma (HCC), cirrhosis (LC), and healthy subjects (HD), and then transplanted the relevant gut microbes into mice that were sterile or free of specific pathogens.
Sequencing of the feces of the three groups of subjects showed that the lowest microbial diversity was found in the feces of patients with hepatocellular carcinoma, but Klebsiella pneumoniae was highly enriched. The gut microbial characteristics of different subjects were very well reproduced in the intestines of the transplanted mice. In addition, a mouse model of induced hepatocellular carcinoma (GFD) based on germ-free mice showed that mice transplanted with hepatocellular carcinoma patients’ intestinal bacteria (HCC-FMT) had the largest tumor diameters, the highest numbers, and the highest burdens.
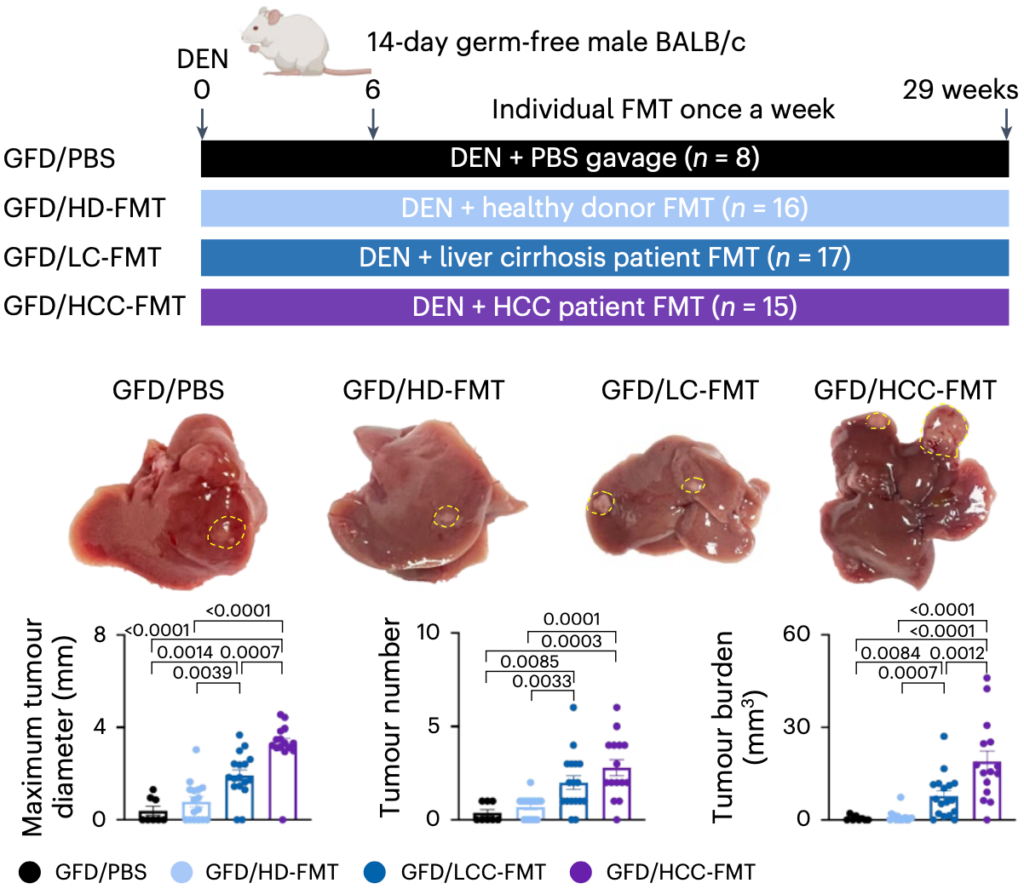
Further assays showed that enterobacteria from transplanted hepatocellular carcinoma patients (HCC-FMT) not only induced hepatocyte proliferation, but also hepatic inflammation, e.g., an increase in pro-inflammatory TH1 and TH17 cells and a decrease in anti-inflammatory TH2 cells.
In addition, gut bacteria from transplanted hepatocellular carcinoma patients also upregulate genes involved in cell proliferation, angiogenesis, embryonic development, epithelial mesenchymal transition, and anti-apoptosis-related genes, as well as increased expression of genes related to pro-inflammatory response and lymphocyte chemotaxis.
Notably, even without the use of the hepatocellular carcinoma inducer diethylnitrosamine (DEN), intestinal bacteria from transplanted hepatocellular carcinoma patients were able to induce the above responses in germ-free mice (GF). Based on this, the researchers concluded that intestinal bacteria from hepatocellular carcinoma patients can spontaneously induce liver inflammation, fibrosis and atypical hyperplasia in mice.
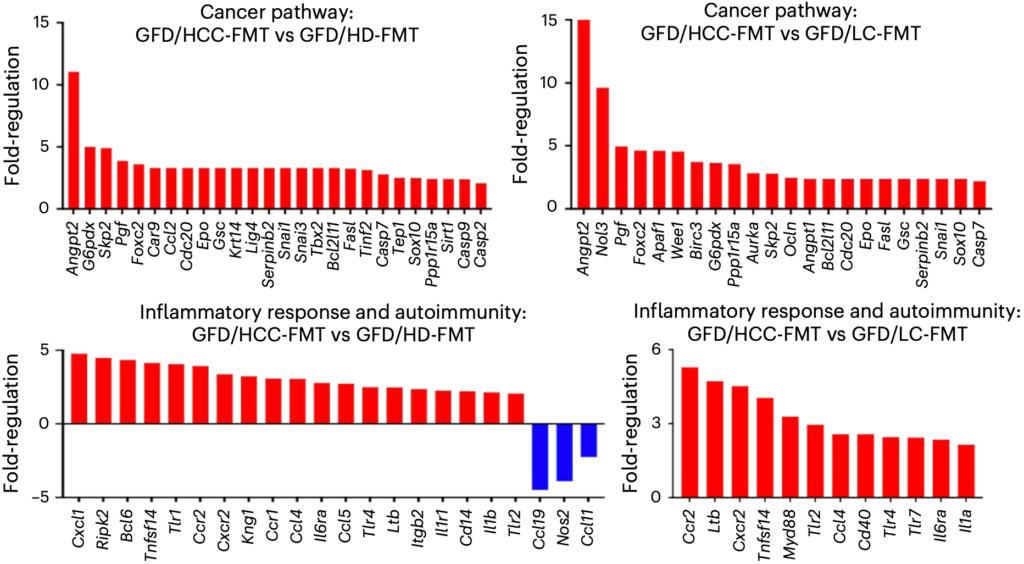
How do enterobacteria get their “hands” on the liver?
Obviously, damage to the intestinal barrier must be the key. Analysis of the results showed that the intestinal permeability of mice transplanted with liver cancer patients’ intestinal bacteria was increased with or without (GFD/GF) carcinogen treatment. Electron microscopic observations showed abnormal intercellular junctions in the mouse colon – loss of tight junctions and widening of adhesions. In addition, there was a decrease in the thickness of colonic mucus, down-regulation of tight junction markers, and greater penetration of bacteria into the lamina propria and muscularis mucosae of the mucosa.
After analyzing mice transplanted with fecal bacteria from different subjects, Jun Yu’s team found that microorganisms were detected in the livers of mice transplanted with fecal bacteria from hepatocellular carcinoma patients, whereas they were almost undetectable in mice transplanted with enterobacteria from healthy subjects. In addition, the most abundant organism in the livers of mice transplanted with fecal bacteria from liver cancer patients was Klebsiella pneumoniae, followed by Enterococcus faecalis, Proteus mirabilis, Enterobacter hormaechei, and Bacteroides fragilis.
After analyzing the liver microbiomes of liver cancer patients, Yu Jun’s team found that Klebsiella pneumoniae was indeed dominant.
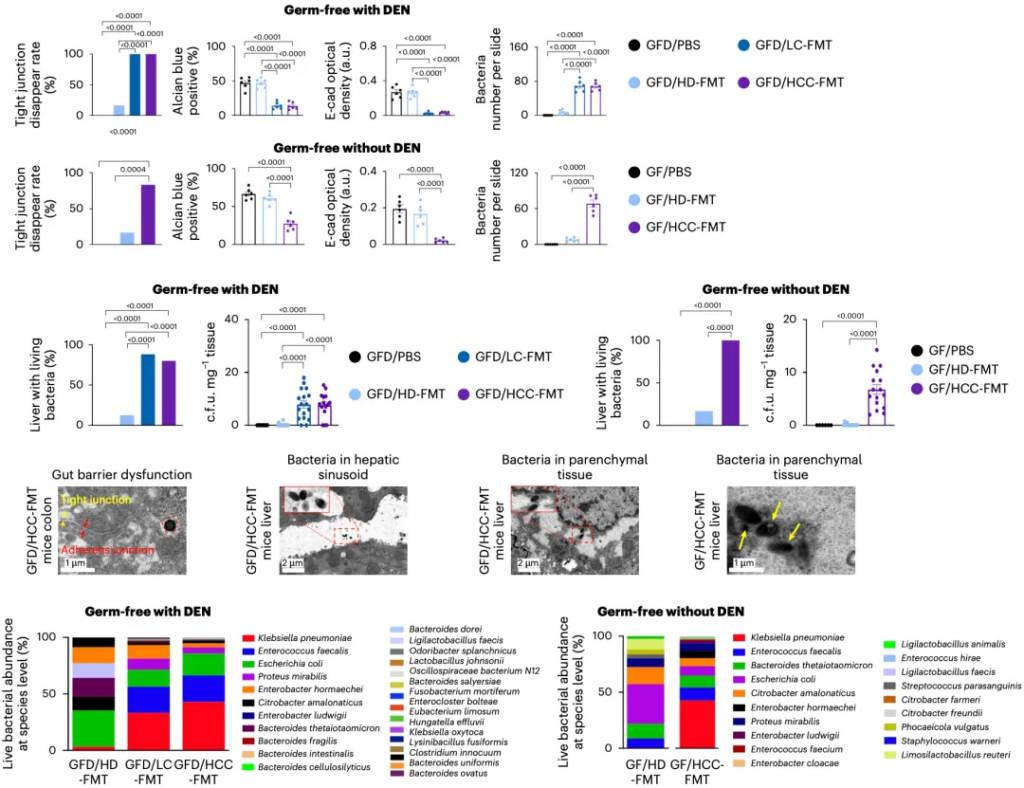
Obviously, the next question was whether gut microbes such as Klebsiella pneumoniae, which traveled thousands of miles from the gut to the liver, were the microbes driving liver cancer.
So, Yu Jun’s team selected five species, including Klebsiella pneumoniae, gram-negative bacteria enriched in the livers of fecal transplanted mice from liver cancer patients, and gave them to sterile mice by gavage. The results showed that only Klebsiella pneumoniae increased intestinal permeability and entered the liver as viable bacteria.
After analyzing the mice’s intestines, they also found that Klebsiella pneumoniae reproduced the detrimental effects of transplanted hepatocellular carcinoma patients’ enterobacteria on the mice’s intestines.
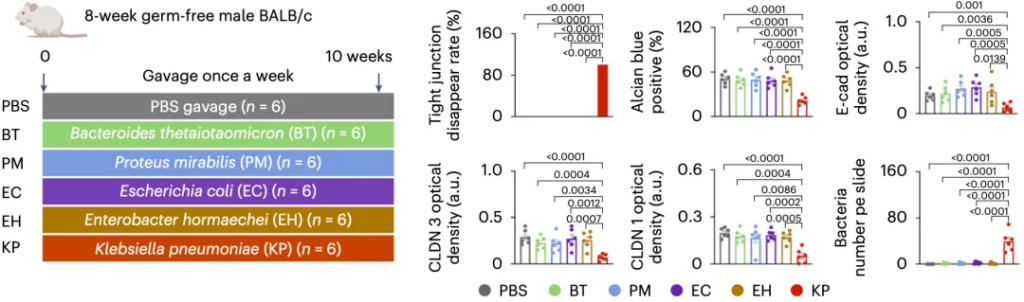
As for the way in which Klebsiella pneumoniae enters the liver, Jun Yu’s team found that Klebsiella pneumoniae promotes the secretion of a gelatinase by intestinal macrophages, which destroys the integrity of the intestinal barrier and then waits for an opportunity to escape from the intestinal tract and enter the liver.
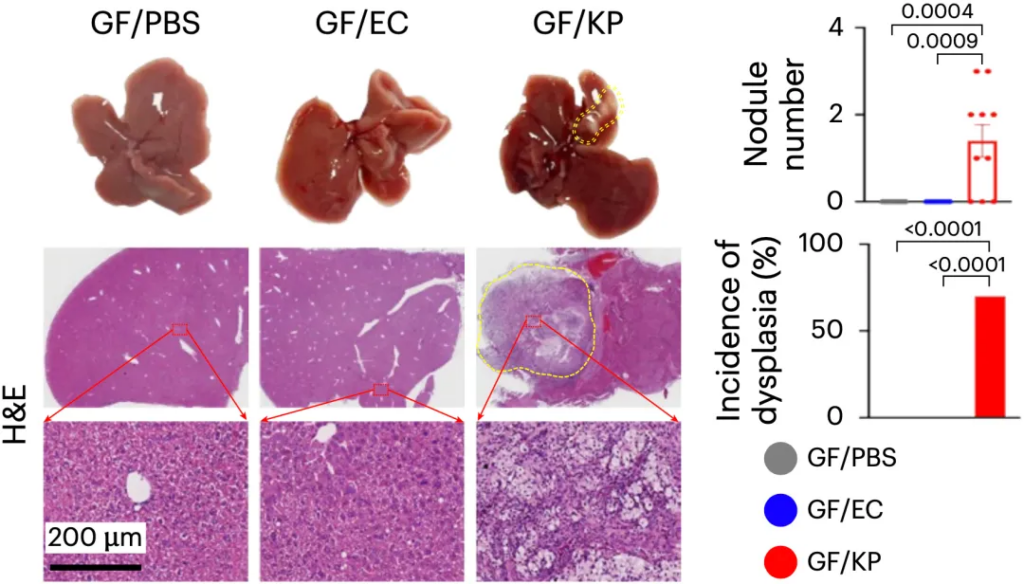
In order to verify whether Klebsiella pneumoniae can cause liver cancer, Jun Yu’s team gavaged sterile mice with Klebsiella pneumoniae for 36 weeks (without carcinogens), and used E. coli and PBS gavage as controls. They found that only Klebsiella pneumoniae induced hepatocyte proliferation, fibrotic damage, oncogenic/pro-inflammatory signaling, and the appearance of atypical hyperplasia (precancerous lesions).
In a mouse model without specific pathogens (SPF), Klebsiella pneumoniae had the same effect. Klebsiella pneumoniae significantly promotes tumor growth if the hepatocellular carcinoma inducer diethylnitrosamine (DEN) is also administered to SPF mice.
Apparently, Klebsiella enterica pneumoniae promotes liver precancerous lesions and hepatocellular carcinoma in mice.
As for the mechanism of Klebsiella pneumoniae promoting hepatocellular carcinoma, Jun Yu’s team found that penicillin-binding protein 1B (PBP1B) in the cell wall of Klebsiella pneumoniae can bind to the receptor TLR4 on the surface of hepatocellular carcinoma cells and activate the TLR4 downstream signaling pathway, which in turn promotes the growth of tumors. And TLR4 inhibitor (TLR4i), TLR4-IN-C34, can inhibit PBP1B-induced liver cancer cell growth.
A mouse model-based study also found that the TLR4 inhibitor TLR4-IN-C34 reversed the effects of Klebsiella pneumoniae on tumor incidence, tumor number and tumor load in DEN-injected germ-free mice. Although TLR4-IN-C34 could not restore the intestinal barrier or prevent Klebsiella pneumoniae from entering the liver, it was effective in inhibiting TLR4 activation, hepatocyte proliferation, fibrosis marker expression, and oncogenic and pro-inflammatory signaling.
Overall, the results of this study by Jun Yu’s team confirm that liver cancer-associated intestinal dysbiosis is causally related to the development of liver cancer, and that Klebsiella pneumoniae is an oncogenic pathogen in liver cancer. This discovery gives us a new knowledge of the relationship between intestinal bacterial imbalance and liver cancer, and also provides new ideas for the prevention and treatment of liver cancer.
According to statistics, the prevalence of intestinal Klebsiella pneumoniae in the population is about 5%, so for this group of people, it is important to protect their intestinal microecology to prevent the enrichment of Klebsiella pneumoniae, or to prevent the development of liver cancer.
Carciclear IgY products, containing immunoglobulins that target FimH adhesin shared by Klebsiella pneumoniae and Escherichia coli, may help maintain a healthy gut microbiome by inhibiting these pathogens.
References
[1].Singh V, Yeoh BS, Chassaing B, et al. Dysregulated Microbial Fermentation of Soluble Fiber Induces Cholestatic Liver Cancer. Cell. 2018;175(3):679-694.e22. doi:10.1016/j.cell.2018.09.004
[2].Wang X, Fang Y, Liang W, et al. Gut-liver translocation of pathogen Klebsiella pneumoniae promotes hepatocellular carcinoma in mice. Nat Microbiol. Published online January 2, 2025. doi:10.1038/s41564-024-01890-9